Q4205
Membrane Wrap™
Dual-Layer Protection for Advanced Wound Healing
A dual-layer amnion-amnion human tissue allograft derived from the amniotic membrane, serving as a barrier and providing protective covering for wounds. Minimally manipulated to preserve natural tissue properties while remaining FDA compliant.
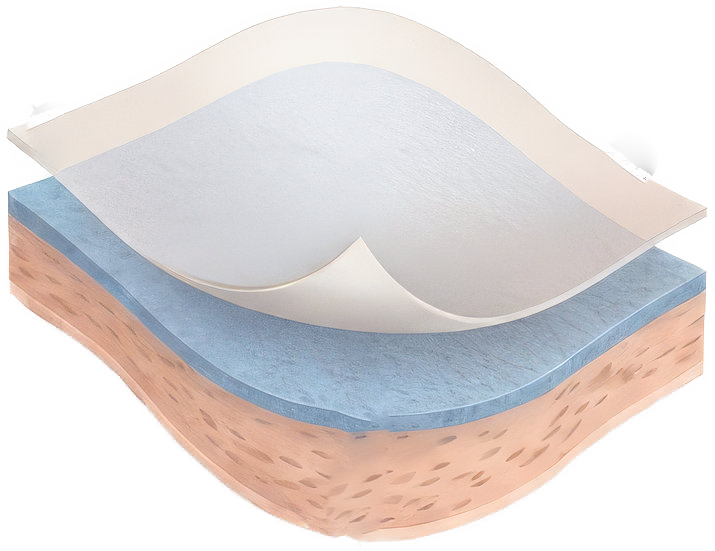
Easy Application. Proven Protection.
Membrane Wrap combines clinical efficacy with operational efficiency, designed for busy practices that demand reliability.
Readily Available
Consistent supply chain ensures product availability when your patients need it most.
Easy Application
Streamlined clinical workflow reduces procedure time and simplifies wound care.
5-Year Shelf Life
Extended shelf life enables excellent inventory management and reduces waste.
FDA Compliant
Manufactured under FDA-compliant processes for regulatory confidence and safety.
Dual-Layer Amnion-Amnion Construction
Membrane Wrap's unique dual-layer structure provides enhanced protection compared to single-layer alternatives.
Primary Amnion Layer
The first amnion layer provides direct wound contact with essential biological properties.
- Natural barrier function
- Growth factors and proteins
- Basement membrane support
- Epithelial layer preservation
Secondary Amnion Layer
The second amnion layer reinforces barrier function and extends protective coverage.
- Enhanced barrier protection
- Additional structural support
- Reinforced wound coverage
- Extended durability
Minimally Manipulated Processing
Membrane Wrap is processed using gentle techniques that preserve the tissue's natural biological properties while ensuring safety and sterility through terminal sterilization.
Clinical Applications
Membrane Wrap provides barrier protection and wound coverage across a range of clinical scenarios.
Wound Protection
- Barrier coverage
- Wound bed preparation
- Protective membrane
Chronic Wounds
- Non-healing wounds
- Venous ulcers
- Diabetic foot ulcers
Surgical Sites
- Post-operative wounds
- Surgical reconstruction
- Donor sites
Acute Wounds
- Partial-thickness wounds
- Soft tissue defects
- Traumatic wounds
Available Sizes
Choose from 5 sizes optimized for common wound dimensions. Simple selection, reduced decision fatigue.
| Size | SKU | Coverage Area |
|---|---|---|
| 2x3 cm | MW0203 | 6 cm² |
| 4x4 cm | MW0404 | 16 cm² |
| 4x6 cm | MW0406 | 24 cm² |
| 4x8 cm | MW0408 | 32 cm² |
| 6x8 cm | MW0608 | 48 cm² |
Safety & Quality Assurance
Membrane Wrap is manufactured under rigorous quality standards to ensure patient safety.
Safety Testing
- Tested for potentially infectious diseases
- Terminally sterilized for each membrane
- FDA Compliant manufacturing
- Minimally manipulated processing
Application Guidelines
Membrane Wrap is designed for streamlined clinical application.
Prepare the wound site
Assess the wound and ensure the wound bed is clean and debrided of necrotic tissue.
Select appropriate size
Choose a Membrane Wrap size that provides adequate coverage with 1-2mm overlap beyond wound margins.
Apply the membrane
Place Membrane Wrap directly on the wound bed. Secure with appropriate secondary dressing.
Heal Faster. Improve Outcomes.
Partner with DermaLynx to bring Membrane Wrap and our complete portfolio of advanced wound care solutions to your practice.